Methylotrophic biomass as 2H-labeled substrate for biosynthesis of inosine
Methylotrophic biomass as 2H-labeled
substrate for biosynthesis of inosine
Oleg V. Mosin1
1 M. V. Lomonosov State Academy of Fine Chemical Technology,
Vernadskogo Prospect 86, Moscow, 117571
Abstract
It was proposed to use the
2H-labeled hydrolysate of RuMP facultative methylotroph Brevibacterium
methylicum, obtained from deuterated salt medium dM9 as a substrate for the
growth of inosine producing bacterium Bacillus subtilis. The growth of
the bacterim was performed via glucose convertion on specially developed medium
dHM with 78.5% (m/m) 2H2O and supplimented with 2.5%
(m/m) of 2H-labeled methylotrophic hydrolysate. To evaluate the
level of deuterium enrichment FAB MS technique was used after the isolation of 2H-labeled
inosine. 2H-labeled inosine obtained from dHM medium represented a
mixture of molecular species containing various number of included deuterium
atoms with different contribution to the enrichment. The level of enrichmet
calculated by the presence of most abandant peak of the molecular ion in
cluster ((M+H)+ at m/z 274) was estimated as five deuterium atoms,
from which three are attributed to ribose and two to hypoxantine.
Keywords: 2H-labeled growth
substrates - Bacillus subtilis - Biosynthesis - 2H-labeled inosine
Introduction
Nucleosides
labeled with deuterium (2H) and other stable isotopes are becoming
an indispensable tool for biomedical diagnostic and the investigation of
various aspects of the metabolism [1, 2]. Thus inosine which is known as an
important intermediate in the synthesis of inosine monophosphate (IMP) is in
the focal point of clinical interest in medical diagnostic of heart deceases
and in certain medical cases [3, 4].
There
are several approaches reported for the preparation of 2H- nucleosides.
Chemical synthesis are usually tedious and inefficient. Only by employing
mutant forms of bacteria, which can produce a large quantities of the
nucleosides when growing of an organism on media containing deuterated
substrates, the desired biochemicals can be obtained both with high yields and
enrichments. On the microbial production of inosine, there have been many
studies so far [5-7]. .
For instance,
a certain adenine, histidine and tyrosine auxotrophic mutants derived from Bacillus
subtilis have been found to have a remarkable ability to produce a large
amount of inosine in the growth medium, and at the present it may be produced
on an industrial scale.
The
major disadvantage of production of 2H-nuclesides is difficulty in
obtaining the appropriate deuterated growth substrates. One approach to solve
this problem is to use the extracts obtained from microorganisms growing on
minimal media with 99,9 at.% 2H2O far [8]. Thus, we
recently described a facultative methylotrophic bacterium Brevibacterium
methylicum, which seems to be an an ideal source for the preparation of
uniformelly labeled growth substrates on the basis of its 2H-biomass
prepared from 2H2O and [U -2H]MetOH [9, 10].
In this article, we demonstrate the possibility of using the hydrolysates of 2H-labeled
biomass of this bacterium as substrates for growing the inosine producing
mutant B. subtillis.
Materials and methods
Chemicals
2H2O
(99.9 at.% 2H[1]) was obtained from
Russian Scientific Enterprises, Sanct Petersburg and purified by distillation
from alkaline permanganate. [U -2H]methanol (95.7 at.% 2H)
was from Biophysic Center, Pushino. All other chemicals were of reagent grade.
To create a high isotopic content in growth medium, 2H2O
with trade marked isotopic purity 99.9 at.% 2H, was used. However,
the deuterium content of used 2H2O verified by NMR was
found to be 97 at.% 2H. The water containing salts were several
times preliminarily crystallyzed in pure 2H2O and dried
in vacuum before using (the true content of deuterium in growth media after the
autoclaving was less smaller on 8-10% then isotopic purity of an initial 2H2O.
The bacterial strain
Adenine, tyrosine and
hystidine auxotroph mutant B. subtilis B -3157 capable to produce and
accumulate 17 g/liter of inosine during the growth on protonated medium with
glucose and yeast extract was employed. The strain was obtained from Russian
State Scientific Center for Genetics and Selection of Industrial Microorganisms
GNIIGENETIKA.
Preparation of 2H-labeled
growth substrates
The methylotrophic
bacterium B. methylicum # 5662 was grown on salt medium dM9 with 93.5%
(m/m) 2H2O and 2% (m/m) [U -2H]MetOH in mass
culture [11]. Cells were pelleted by centrifugation (2000 g, 10 min), washed
once with 2H2O and stored at -14 0C.
Periodically, 10 g (wet weight) portions are thawed, suspended in 0.5 N 2HCl
solution (in 2H2O) and autoclaved at 1200C for
30 min. After adjusting pH till 7.0-7.2 with potassium hydroxide, the
hydrolysate was used as a mixure of 2H-labeled growth substrates for
the growth of inosine producing strain.
Media and growth
conditions
The bacterial growth was
carried out on FM medium (m/m.%): glucose 12; yeast extract 2.5; ammonium
nitrate 3; magnium sulphate 2; chalk 2. The composition of dHM was as the same
as FM except dHM was prepared from 2H2O and the
hydrolysate of 2H-labeled methylotrophic biomass was added. The
media were sterilized by autoclaving at 1200C for 30 min and cooled.
Glucose was sterilized separetely in 2H2O solution, and
after that added in growth medium. рН was adjusted till 6.5-6.7 with potassium hydroxide. The bacterium was
grown in 250 ml Erlenmeyer flasks containing 20 ml of the medium at 32-34 0С and vigorously aerated on an orbital
shaker. After 7 days the cells were pelleted by centrifugation (2000 g, 10
min). The supernatant was separated, lyophilized and used for the isolation of 2H-labeled
inosine.
Isolation of inosine
MetOH solution in H2O
(50 v/v %, 20 ml) was added to a lyophilized growth medium. The mixture was
allowed to - 4 0C and after 10 h the total protein was precipitated
and removed by centrifugation (1200 g, 10 min). MetOH was evaporated under
reduced pressure. The resulting mixture was dissolved in 2H2O
(30 ml) and 5 g of activated carbon was added. After keeping for 24 h at -4 0C,
the inosine, eluting with ammonia, was concentrated and twice recrystallized
from MetOH (nd20 = 1.33). The purity of the product was
judged by using controls of normal nucleosides, and running mixed TLC with
graded amounts of the neighboring nucleosides.
Quantitative determination
During the growth inosine
was separated by TLC on Silufol UV-254 plates with mobile phases: n -ButOH -
AcOH - water (2:1:1, v/v) using pure commercial available inosine as a
standard. The amount of inosine was determined for 10 ml aliquots of liquid
growth medium by TLC. The sports were eluted by 0.1 N solution of HCl (10 ml).
The absorbance of the eluates was measured at 249 nm and the content of inosine
was determined using a standard curve.
The convertion of glucose
was estimated enzymatically with glucoseoxydenase method [].
Equipment
Absorbance was measured
with a spectrophotometer Beckman DU-6 (USA).
The analysis of protein
hydrolisates was carried out using a Biotronic LC 50001 chromatograph
(Germany), 230 x 3.2 mm, working pressure 50-60 atm, flow-rate 18.5 ml/h.
The levels of deuterium
enrichment of amino acids were investigated with the aid of EI MS after
derivatization to methyl esters of N-Dns-amino acids [].
FAB MS was performed on
Hitachi MBA spectrometer (Japan) on glyserol template at potential 5 кV and an ion current of 0.6-0.8 мА.
RESULTS AND DISCUSSION
Production of 2H-labeled
inosine
For biosynthesis of 2H-
labeled inosine we employed bacterium Bacillus subtillis, which
could produce and accumulate a conciderable amount of inosine exogeniously due
to an altered nucleoside metabolism. This strain displayed the maximum
productivity on FM medium, containing as a source of carbon and energy glucose
(12 m/m.%), and as a source of growth factors and additional source of nitrogen
the yeast extract. Since the small availability of commercial available 2H-labeled
biomass prepared from yeast, it was necessary to find the more suitable
microbial source, from which the 2H-labeled growth substrates could
be obtained. For this purpose we employed the available RuMP facultative
methylotroph Brevibacterium methylicum [5] with the content of the total
protein and polycarbohydrates in biomass 53 and 10% respectively [6].
The content of
amino acids in biomass of B. methylicum and the deuterium enrichment are
shown in Table.
Table:
The content of amino acids in biomass of B.
methylicum and the deuterium enrichment.
|
Amino Acids
|
The content in biomass, %
|
Deuterium enrichment , %
|
|
Glycine
|
9,69
|
90,0
|
|
Alanine
|
13,98
|
97,5
|
|
Valine
|
3,74
|
50,0
|
|
Leucine/Isoleucine
|
7,33/3,64
|
49,0
|
|
Phenylalanine
|
3,94
|
95,0
|
|
Tyrosine
|
1,82
|
92,8
|
|
Serine
|
4,90
|
90,0
|
|
Threonine
|
5,51
|
not determined
|
|
Methionine
|
not determined
|
|
Aspartic Acid
|
9,59
|
66,6
|
|
Glytamic Acid
|
10,38
|
70,0
|
|
Lysine
|
3,98
|
58,9
|
|
Arginine
|
5,27
|
not determined
|
|
Histidine
|
3,72
|
not determined
|
The hydrolysis of 2H-labeled
biomass was performed in mild conditions via its autoclaving (30 min, 08 atm)
in 0.5 N solution of 2HCl (in 2H2O). The data
on the amino acid composition of hydrolysate and levels of the enrichment are
shown in Fig. 2. The contents of tyrosine and histidine in hydrolysate were
1.82 and 3.72% and can ensure the polyauxotrophy of the inosine producing
strain. Another important parameter is a high level of amino acid enrichment.
Bacterial growth and
production of inosine
Two following media were
used for the bacterial growth:
1). FM medium, prepared from ordinary
protonated water and yeast extract.
2). dHM medium, prepared from 87.5%
(v/v) 2H2O and 2.5% (m/m) of 2H-labeled
methylotrophic hydrolisate, obtained accordingly from medium dМ9.
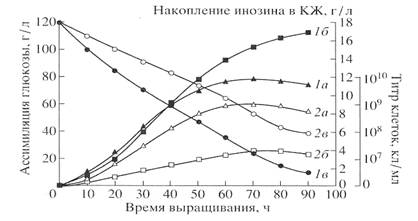
Fig.1
Curves, reflecting the
growth dynamics (a), convercion of glucose (b) and production of inosine (c)
are given in Fig. 1. A maximal level of inosine production on ordinary
protonated medium was 17 g\liter. When growing on dHM medium the strain
produced only 3.9 g/liter of inosine throughout the whole course of the growth.
The low level of inosine production was correlated with a degree of glucose
conversion in those conditions. 4m/m.% of non-assimilated glucose was detected
in medium dHM after the growth, that proved that glucose is metabolized less
effectivelly on medium dHM, that is probably a result of non-equvalent
replacement of yeast extract by methylotrophic hydrolysate.
The absorption
spectra of inosine isolated from medium dHM (a) are shown in Fig. 2
comparatively to the growth medium (b) and commertially available inosine (c).
TLC of isolated inosine showed the presence of main spot with Rf=0.5
(inosine) and additional spot with Rf=0.75 (hypoxantine). The output
of 2H-labeled inosine was 1 gram from 1 liter of growth medium.
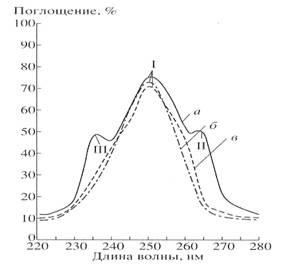
Fig.2
The evaluation of inosine
enrichment
The method of FAB MS was
employed for the evaluation of inosine enrichment. The fragmentation pathways
of inosine by FAB MS are shown in Fig. 3. Two main decomposition processes
arised from the molecule: sugar (m/z 133) and hypoxantine (m/z 136) formation.
The compounds with a smaller m/z ratio may further to be formed as a result of
elitination of HCN and CO from hypoxantine. The level of deuterium enrichment
could be evaluated from the FAB mass spectrum of 2H-labeled inosine
shown in Fig. 3, b compared with the non-labeled inosine (a). The results,
firmely established the labeling of inosine as heterogenious, juging by the
presence of clasters of adduct peaks at the molecular ion MH+; the
species of molecules with different numbers of deuterium atoms were visualised.
The most abundant peak with (M+H)+ at m/z 274 (instead of m/z 269
for non-labeled compound) in the claster was registered by mass spectrometer as
a peak with average m/z ratio, from whom the enrichment of inosine was
calculated as five deuterium atoms. The presence of peak corresponding to the
hypoxantine fragment [C5H4ON4]+ at m/z 138
(instead of m/z 136) and the peak of sugar fragment [C5H9O4]+
at m /z 136 (instead of m/z 133) proved that two deuterium atoms are located in
hypoxantine, however, three of them are attributed to the ribose pattern.
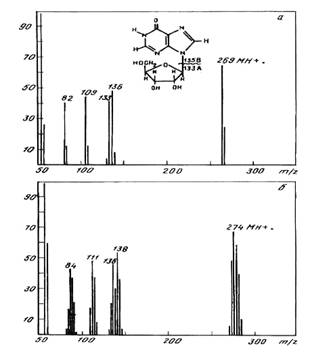
Fig.3
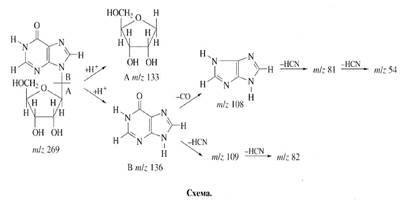
Mainly two aspects of
the enrichment of inosine were taken into account (scheme). First, because
protons in С’1-С’5 positions of ribose pattern in
inosine could be originated from glucose, we assumed, that the character of
biosynthetic enrichment of deuterium in sugar pattern of inosine is determined
mainly to the functioning of a number of processes of hexose monophosphate
shunt of glucose assimilation. But since protonated glucose was added in growth
medium, its contribution in the inosine enrichment was minimal. Nevertheless,
the results suggested, that ribose contained three deuterium atoms that could
not stemp from glucose. Three deuterium atoms probably stemp via some minor
reactions of glucose biosynthesis. Secondly, the numerous exchange processes
and intermolecular regrouping reactions, occurring with participation of 2H2O
could also be resulted in specific labelling of inosine. Such accessible
positions are occupied by the easily exchangeable hydrogen (deuterium) atoms
both of hydroxylic- and imino groups of inosine. Two protons at C-H positions
in inosine could be replaced by deuterium via assimilation of 2H-labeled
hydrolysate. The enrichment of inosine was approximately the same as 2H2O
content in growth medium (65.5-67.5%).
LITERATURE.
1. Munch-Petersen A., (1983)
Metabolism of nucleotides, nucleosides, and nucleobases in microorganisms.
Academic Press. Inc., New York. 105.
2. Wuthrich K. (1986) NMR of proteins
and nucleic acids. New York: J. Wiley & Sons. 14.
3. Bloch A. (1975) Chemistry,
biology, and clinical uses of nucleoside analogs. Academic Press, New York. 58.
4. Farber E., Shull H., McConomy
J.M., and Castillo A.E. (1965) Biochem. Pharmacol. 14, 761.
5. V.I. Shvets, A.M. Yurkevich, O.V.
Mosin, D.A. Skladnev. (1995) Karadeniz Journal of Medical Sciences. 8.
No 4. P.231-232.
6. Ishii K., & Shiio I., (1972) Agric.
Biol. Chem. 36, 1511-1522.
7. Matsui H., Sato K., Enei H., and
Takinamy K., (1982) Agric. Biol. Chem. 46, 2347-2352.
8. Katz J. & Crespi H.L. (1972)
Pure Appl. Chem. 32, 221-250.
9. Mosin O.V., Karnaukhova E.N.,
Pshenichnikova A.B., et al. (1993) Biotechnology (Russia). 9, 16-20.
10. Egorova T.A., Mosin O.V., Eremin
S.V., Karnaukhova E.N., Zvonkova E. N., Shvets V.I. 91993) Biotechnology
(Russia) 8, 21-25.