The new method of gas saturated oil viscosity determination
The New Method of Gas Saturated
Oil Viscosity Determination
V.I., Krikunov V.V.paper presents a
mathematic technique for calculating viscosity of West Siberian reservoir
oils.The viscosity is one of the most important fluids parameters, is submitted
as a function of viscosities of components included in gas saturated oil,
reservoir temperature and pressure.method uses concept of standard viscosity
reservoir oil (at 200С and 0.1 МPа) - hypothetical values, since the mix at
these conditions is double phase. The influence of temperature and pressure on
standard viscosity has been defined by correlations obtained at research of
behavior of West Siberian subsurface collected oil samples.laboratory practice
of crude oil saturated with natural gas dynamic viscosity study in reservoir
conditions high-pressure viscometers, which is included as additional components
in complex and power-intensive PVT-equipment simulating reservoir pressure and
temperature are used. In conditions of the analysis of the representative
series of samples that show behavior of reservoir fluids, the realization of
the large number of definitions is required that causes a lot of difficulties.
Use of only experimental methods is connected to the time-consuming for
realization of the analysis, presence of precision devices in laboratory,
necessity of continuous operator observing for run, etc. Besides the factors
listed above, there are a number of restrictions in using one or others
viscometers depending on viscosity of a sample has been under study [1].
Therefore creation of new calculation methods of the basic characteristics of
oil-gas mixes used for oil fields development and surface facilities
construction has both scientific and practical importance.development of a
calculation method of dynamic viscosity determination of mixture its
composition is chosen as initial parameter because it gives the best
description of a mixture. The knowing of apparent viscosity of pure components
at standard conditions is initial parameters for the method. Briefly, after
composition as a whole has been analyzed followed by using the appropriate
mixing rule for the apparent viscosity of a crude oil-gas mixture is defined.
For further steps, the method uses the empirical correlations of standard
viscosity, pressure and temperature for determination of oil saturated with gas
viscosity at reservoir conditions.
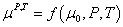 (1)
(1)
where: μP,
T dynamic viscosity of hydrocarbon fluid at P,T, mPa·s;
μ0
apparent viscosity of hydrocarbon fluid at standard conditions (0.1MPa, 293K),
mPa·s, (standard viscosity)., the standard viscosity can be presented as simply
combination of definitively functions of pure component standard viscosity
according to its molar fraction. For example, in earlier published works the
combination of additive viscosity function 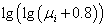 of cut fractions for tank oil takes
place or the equation for gases at low pressure depending on molar composition
is used:
of cut fractions for tank oil takes
place or the equation for gases at low pressure depending on molar composition
is used:
|
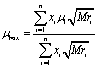 (2) (2)
|
|
Experimental Data Obtained
In process of obtaining experimental
data the viscosities of reservoir liquid were obtained. The data were in
temperature and pressure range 50-900C and 17-32 MPa. The way of experimental
definition of viscosity consists of follow steps: sample of reservoir mixture
has been transferred by non-mercury displacement RUSKA from first equilibrium
cell into second cell thru the calibrated capillary of a known diameter at
constant temperature (reservoir temperature) and fixed pressure difference,
that equal to a difference between reservoir and some pressure that is bellow
than reservoir, but higher than saturation point pressure. Composition of
mixture has been calculated by means of result of gas chromatography analysis
of liberated phases (gas and oil) and gas/oil ratio at standard (at 200C,
0.1MPa) flash separation.
Development of the Calculation
Procedures
Apparent Viscosity at Standard
Conditionsdefinition of standard viscosity of oil-gas mixture we propose the
following mixing rule:
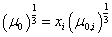 (3)
(3)
Earlier the
similar equation was used by Kendall and Monroer for definition of binary mixes
viscosity depending on value of viscosity of components included in their
composition [2]. In equation (3): xi is a molar fraction
of i component; μ0,i is a standard viscosity of i component. The values of
μ0,i for natural mixed components are presented in Table 1.
1 -
Initial values of liquid density and dynamic viscosity of crude oil-gas mixes
components at 200C and 0.1 MPa
|
Component
|
Molar weight
|
Liquid Density, kg/m3
|
Liquid Viscosity, mPa·s
|
|
Carbon dioxide………………….
|
44.01
|
850
|
0.26
|
|
Nitrogen………………………...
|
28.02
|
570
|
0.12
|
|
Methane………………………...
|
16.04
|
(300)
|
(0.036)
|
|
Ethane…………………………..
|
30.07
|
(460)
|
(0.086)
|
|
Propane…………………………
|
44.09
|
501
|
0.120
|
|
Isobutane………………………..
|
58.12
|
557
|
0.179
|
|
Norm. butane…………………...
|
58.12
|
580
|
0.171
|
|
Isopentane………………………
|
72.14
|
620
|
0.224
|
|
Norm. pentane………………….
|
72.14
|
626
|
0.234
|
of components, since pentane,
correspond to their actual value at standard conditions (Reference
Data).components C3 and C4 in a mix have properties similar to normal liquid.
The values of apparent viscosity for them are offered to be defined by
extrapolation of the equation Chatteree, Kunte for organic liquids [3].
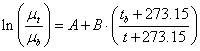 (4)
(4)
where: μt-
viscosity of a liquid at given temperature, mPa·s
μb
- viscosity of a liquid at boiling temperature, mPa·s- boiling temperature, 0C
- current value of temperature;
А =-2.6677;
B = 2.6613 (for paraffin hydrocarbons)values of apparent viscosity for methane
and ethane are received by using of following expressions (Starling-Ellington
methods [5]):
|
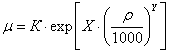 ,(5) ,(5)
|
|
: Y, К, Х - Lee coefficients adapted
for temperature in Celsius degrees:
t - temperature, 0C;- molecular
weight of component;
ρ - liquid density of component,
kg/m3 (at 200C - apparent liquid density).determination of apparent density
methane and ethane in the mixture Standing correlation expressions is applied
[4]:
|
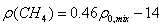 ,(5) ,(5)
|
|
|
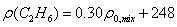
|
|
Where: ρ0,mix - apparent density of
mixture that is computed by the following equation (Standing):
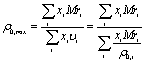 (6)
(6)
Where: υi -
molar volume of i component.
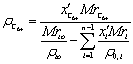 (7)
(7)
Where: Mrto
- cryoscopy molar weight of tank oil;
ρ0 -
density of tank oil;
x′i -
molar fraction of i component in tank oil;- molar weight of i component from
Table 1. For С6+-fraction the value of molar weight obtained from following
expression:
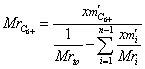 (8)
(8)
Where: xm′i
- GC mass fraction of i component in tank oil., knowing of apparent density of
a mixture it is possible to define density of methane and ethane with the
equation (5). The final value of C1, C2 density is found out by Newton’s
successive approximation method with convergence criterion 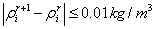 , where r is
number of iteration. 3 or 4 approximations are enough for realization of the
given algorithm. The apparent viscosity of methane and ethane is specified
their density. Taken magnitude of C1, C2 viscosity is subsequently used for of
oil-gas mixture apparent viscosity estimation. С6+-fraction the value of
viscosity has been obtained by expression (3) rewritten for C6+ viscosity in
tank oil:
, where r is
number of iteration. 3 or 4 approximations are enough for realization of the
given algorithm. The apparent viscosity of methane and ethane is specified
their density. Taken magnitude of C1, C2 viscosity is subsequently used for of
oil-gas mixture apparent viscosity estimation. С6+-fraction the value of
viscosity has been obtained by expression (3) rewritten for C6+ viscosity in
tank oil:
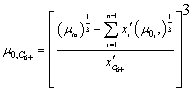 (9)
(9)
Where: μ0 -
viscosity of tank oil.
Temperature
and Pressure Influence
As was
earlier shown the viscosity of oil-gas system at reservoir conditions depends
on given pressure, temperatures and standard viscosity. The kind of functional
dependence such as (1) is received from a study more than 250 bottom-hole
samples, which was collected from more than 80 West Siberian oil fields. It has
been found out that viscosity is completely described by the following
function:
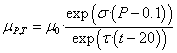 (10)
(10)
Where: σ
and τ - baric and thermal coefficients of viscosity, respectively.expressions
for definition σ and τ depending on molar weight of reservoir mixture
(rm) have sufficient prediction of accuracy for West Siberian oil-gas mixes and
are following:
|
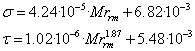 ,(11) ,(11)
|
|
Use of the procedure
Table 2 summarized the results of
bottom-hole study of typical West Siberian reservoir oil. Fig.1 shows the
result of application of the method with experimental data for comparison. The
average absolute deviation of these calculated and experimental viscosities is
11.1%. Thus some statistical increase of an error with increase of pressure and
temperature is observed. The absolute value of standard viscosity practically
has not influence on value of an error. It is comparable with errors of another
known methods, but the basic rules of proposed method are justified in terms of
physical chemistry and the calculations are easier realized. For these reasons
it is possible to consider this method satisfactory for calculation of
viscosity of West Siberian crude oil saturated with natural gas.
oil gas crude
Table 2
- Chart of experimental conditions and properties
of crude oil saturated with natural gas samples
|
# of sample
|
Pressure, MPa
|
Temperature, 0C
|
0
|
Mrrm
|
|
|
Experimental
|
Calculated
|
r,%
|
|
1
|
19.23
|
69
|
1.531
|
112
|
0.012
|
0.012
|
1.04
|
1.04
|
0.00%
|
|
2
|
19.14
|
69
|
1.518
|
112
|
0.012
|
0.012
|
1.04
|
1.03
|
0.96%
|
|
3
|
29.12
|
90
|
1.623
|
129
|
0.015
|
0.012
|
1.15
|
0.84
|
26.96%
|
|
4
|
21.65
|
69
|
1.636
|
107
|
0.012
|
0.011
|
1.35
|
1.17
|
13.33%
|
|
5
|
17.1
|
65
|
121
|
0.013
|
0.012
|
1.18
|
1.22
|
-3.39%
|
|
6
|
16.38
|
67
|
2.585
|
140
|
0.016
|
0.013
|
1.36
|
1.5
|
-10.29%
|
|
7
|
23.69
|
67
|
4.152
|
116
|
0.013
|
0.012
|
2.98
|
2.99
|
-0.34%
|
|
8
|
21.47
|
61
|
5.061
|
200
|
0.026
|
0.015
|
2.45
|
2.42
|
1.22%
|
|
9
|
25.1
|
70
|
5.529
|
156
|
0.018
|
0.013
|
2.42
|
3.09
|
-27.69%
|
|
10
|
14.86
|
56
|
6.915
|
159
|
0.019
|
0.014
|
3.95
|
4.29
|
-8.61%
|
|
11
|
23.45
|
73
|
7.224
|
160
|
0.019
|
0.014
|
4.68
|
3.63
|
22.44%
|
|
12
|
19.16
|
67
|
8.444
|
0.021
|
0.014
|
5.74
|
4.19
|
27.00%
|
|
13
|
19.44
|
67
|
9.048
|
175
|
0.021
|
0.014
|
4.2
|
4.35
|
-3.57%
|
|
14
|
17.77
|
55
|
9.126
|
183
|
0.023
|
0.015
|
5.28
|
5.31
|
-0.57%
|
|
15
|
18.81
|
65
|
9.371
|
196
|
0.025
|
0.015
|
4.21
|
4
|
4.99%
|
|
16
|
24.73
|
70
|
9.418
|
170
|
0.021
|
0.014
|
4.81
|
4.75
|
1.25%
|
|
17
|
18.67
|
68
|
9.674
|
179
|
0.022
|
0.014
|
4.28
|
4.37
|
-2.10%
|
|
18
|
22
|
69
|
10.375
|
182
|
0.023
|
0.015
|
3.86
|
4.7
|
-21.76%
|
|
19
|
24.63
|
69
|
15.025
|
180
|
0.014
|
8.1
|
7.18
|
11.36%
|
|
20
|
27.91
|
67
|
15.436
|
194
|
0.025
|
0.015
|
6.4
|
7.3
|
-14.06%
|
|
21
|
27.83
|
82
|
17.296
|
179
|
0.022
|
0.014
|
5.76
|
6.54
|
-13.54%
|
|
22
|
19.92
|
59
|
24.295
|
212
|
0.028
|
0.016
|
9.56
|
11.01
|
-15.17%
|
|
23
|
28.13
|
84
|
33.183
|
188
|
0.024
|
0.015
|
14
|
11
|
21.43%
|
|
24
|
20.41
|
59
|
33.369
|
212
|
0.028
|
0.016
|
13.57
|
15.24
|
-12.31%
|
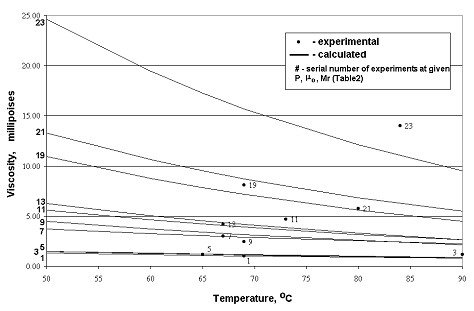 1 - Calculated and
Experimental Results Comparison
1 - Calculated and
Experimental Results Comparison
Reference
1. Fuks
G.I. (1951) Viscosity and Plasticity of Petroleum, p.270.
2. Victorov
M.M. (1977) Method of calculation of physical and chemical values and applied
accounts, p.263.
. Chatteree
A., Kunte A.V. (1982) Estimation of Viscosity of Organic Liquids, Chemistry
& Industry, 11: 375-376.
. Shilov
V.I. (1981): Density Determination of gas Saturated Oil and Oil-Water Mixes,
Oil Industry, 3:58.
. Starling
K.E., Ellington R.T. (1964) Viscosity Correlation for Nonpolar Dense Fluids,
A.I.Ch.E. Journal, 1:11 - 15.