Ecological problems. Environmental protection
Министерство
образования и науки Украины
Днепропетровский
областной медицинский
Лицей –
интернат «Днiпро»
Курсовая
работа
«Ecological problems.
Environmental protection»
Выполнила:
Ученица 11 – Г класса
Калашникова Анастасия
Научные руководители:
Кривонос И.А.
Легкий П.В.
Introduction
My term-paper
is devoted to the theme of the global ecological problems and the environmental
protection. I would like to tell you about some problems for example
“Greenhouse effect”. The aim of my project is to show and explain how
ecological problems influence on our life and about there consequences.
The sources of
my work are:
·
Scientific
books and newspapers
·
Numerous
internet data
My project
consists of the following parts: Introduction, Literature overview, conclusion
and literature.
·
Literature
overview consists of 11 themes.
·
Conclusion.
·
Literature.
I suppose that the topic I chose is
very actual nowadays and I hope that it will contribute to our knowledge and
will also have a practical implementation in the class.
Ecological situation nowadays
Ecology is a
very popular word today. But what does it mean? Ecology is a since which
studies the relationship between all forms of life on our planet and the
environment. This word came from Greek “oikos” which means home. The idea of
home includes our whole planet, its population, Nature, animals, birds, fish,
insects and all other living beings and even the atmosphere around our planet.
Since ancient times Nature has served Man
giving everything he needs: air to breathe, food to eat, water to drink, wood
for building and fuel for heating his home. For thousands of years people lived
in harmony with the environment and it seemed to them that the resources of
nature had no end or limit. With the industrial revolution our negative
influence on Nature began to increase. Large cities with thousands of steaming,
polluting plants and factories can be found nowadays all over the world. The
by-products of their activity pollute the air we breathe the water we drink the
fields where our crops are grown. That’s why those who live in cities prefer
spending their days off and their holidays far from the noise of the city, to
be closer to nature. Perhaps they like to breathe fresh air or to swim in clear
water because the ecology is not so poor as in the cities.
So, pollution is one of the most burning
problems of nowadays. Now millions of chimneys, cars, buses, trucks all over
the world exhaust fumes and harmful substances into the atmosphere. These
poisoned substances pollute everything: air, land, water, birds and animals.
So, it is usually hard to breathe in the large cities where there are lots
plants.
Every year the atmosphere is polluted by
about 1000 tons of industrial dust and other harmful substances. Big cities
suffer from smog. Cars with their engine have become the main source of
pollution in industrial countries. Vast forests are being cut down for the need
of industries in Europe and USA. The loss of the forests upsets the oxygen
balance of the new wastelands. As the result some species of animals, birds,
fish and plants have disappeared and keep disappearing.
Water pollution is very serious, too. Ugly
rivers of dirty water polluted with factory waste, poisoned fish are all-round
us. And polluted air and poisoned water lead to the end of the civilization.
So, nowadays a lot of dead lands and lifeless areas have appeared, because our
actions and dealings can turn the land to a desert.
Greenhouse
effect
The greenhouse
effect is the process in which the emission of infrared radiation by the
atmosphere warms a planet's surface. The name comes from an analogy with the
warming of air inside a greenhouse compared to the air outside the greenhouse.
The Earth's average surface temperature is about 33°C warmer than it would be without the greenhouse effect. The greenhouse effect was discovered by
Joseph Fourier in 1829 and first investigated quantitatively by Svante
Arrhenius in 1896. In addition to the Earth, Mars and especially Venus have
greenhouse effects.
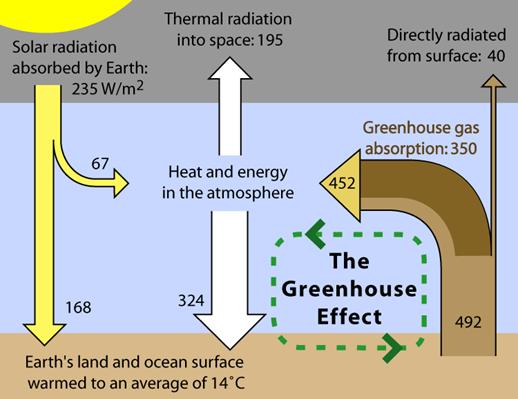

Basic
mechanism
ecological
environmental protection greenhouse
The Earth
receives energy from the Sun in the form of radiation. The Earth reflects about
30% of the incoming solar radiation. The remaining 70% is absorbed, warming the
land, atmosphere and oceans. For the Earth's temperature to be in steady state
so that the Earth does not rapidly heat or cool, this absorbed solar radiation
must be very nearly balanced by energy radiated back to space in the infrared
wavelengths. Since the intensity of infrared radiation increases with
increasing temperature, one can think of the Earth's temperature as being
determined by the infrared flux needed to balance the absorbed solar flux. The
visible solar radiation mostly heats the surface, not the atmosphere, whereas
most of the infrared radiation escaping to space is emitted from the upper
atmosphere, not the surface. The infrared photons emitted by the surface are
mostly absorbed in the atmosphere by greenhouse gases and clouds and do not
escape directly to space.
The reason
this warms the surface is most easily understood by starting with a simplified
model of a purely radiative greenhouse effect that ignores energy transfer in
the atmosphere by convection (sensible heat transport) and by the evaporation
and condensation of water vapor (latent heat transport). In this purely radiative
case, one can think of the atmosphere as emitting infrared radiation both
upwards and downwards. The upward infrared flux emitted by the surface must
balance not only the absorbed solar flux but also this downward infrared flux
emitted by the atmosphere. The surface temperature will rise until it generates
thermal radiation equivalent to the sum of the incoming solar and infrared
radiation.
A more
realistic picture taking into account the convective and latent heat fluxes is
somewhat more complex. But the following simple model captures the essence. The
starting point is to note that the opacity of the atmosphere to infrared
radiation determines the height in the atmosphere from which most of the
photons are emitted into space. If the atmosphere is more opaque, the typical
photon escaping to space will be emitted from higher in the atmosphere, because
one then has to go to higher altitudes to see out to space in the infrared.
Since the emission of infrared radiation is a function of temperature, it is
the temperature of the atmosphere at this emission level that is effectively
determined by the requirement that the emitted flux balance the absorbed solar
flux.
But the
temperature of the atmosphere generally decreases with height above the
surface, at a rate of roughly 6.5 °C per kilometer on average, until one reaches the stratosphere 10-15 km above the surface. (Most infrared photons escaping
to space are emitted by the troposphere, the region bounded by the surface and
the stratosphere, so we can ignore the stratosphere in this simple picture.) A
very simple model, but one that proves to be remarkably useful, involves the
assumption that this temperature profile is simply fixed, by the non-radiative
energy fluxes. Given the temperature at the emission level of the infrared flux
escaping to space, one then computes the surface temperature by increasing
temperature at the rate of 6.5 °C per kilometer, the environmental lapse rate, until one reaches the surface. The more opaque the atmosphere, and the
higher the emission level of the escaping infrared radiation, the warmer the
surface, since one then needs to follow this lapse rate over a larger distance in
the vertical. While less intuitive than the purely radiative greenhouse effect,
this less familiar radiative-convective picture is the starting point for most
discussions of the greenhouse effect in the climate modeling literature.
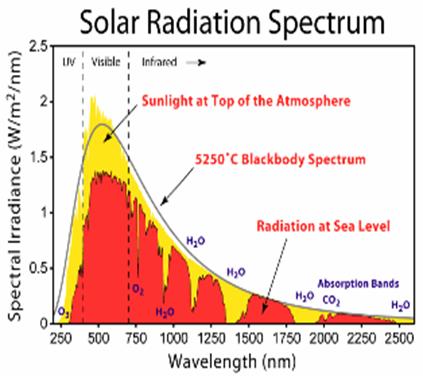
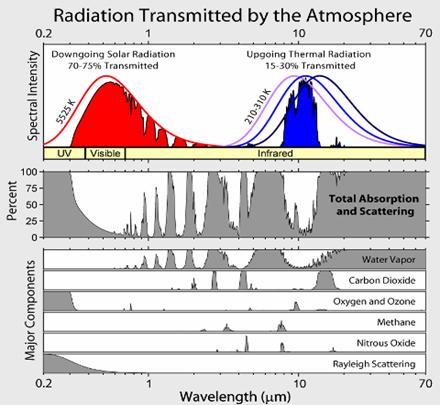
Greenhouse
gases
Quantum
mechanics provides the basis for computing the interactions between molecules
and radiation. Most of this interaction occurs when the frequency of the
radiation closely matches that of the spectral lines of the molecule,
determined by the quantization of the modes of vibration and rotation of the
molecule. (The electronic excitations are generally not relevant for infrared
radiation, as they require energy larger than that in an infrared photon.)
The width of a
spectral line is an important element in understanding its importance for the
absorption of radiation. In the Earth’s atmosphere these spectral widths are
primarily determined by “pressure broadening”, which is the distortion of the
spectrum due to the collision with another molecule. Most of the infrared
absorption in the atmosphere can be thought of as occurring while two molecules
are colliding. The absorption due to a photon interacting with a lone molecule
is relatively small. This three-body aspect of the problem, one photon and two
molecules, makes direct quantum mechanical computation for molecules of
interest more challenging. Careful laboratory spectroscopic measurements
provide the basis for most of the radioactive transfer calculations used in
studies of the atmosphere.
The
molecules/atoms that constitute the bulk of the atmosphere: oxygen (O2),
nitrogen (N2) and argon; do not interact with infrared radiation
significantly. While the oxygen and nitrogen molecules can vibrate, because of
their symmetry these vibrations do not create any transient charge separation.
Without such a transient dipole moment, they can neither absorb nor emit
infrared radiation. In the Earth’s atmosphere, the dominant infrared absorbing
gases are water vapor, carbon dioxide, and ozone (O3). The same
molecules are also the dominant infrared emitting molecules. CO2 and
O3 have "floppy" vibration motions whose quantum states
can be excited by collisions at energies encountered in the atmosphere. For
example, carbon dioxide is a linear molecule, but it has an important
vibrational mode in which the molecule bends with the carbon in the middle
moving one way and the oxygens on the ends moving the other way, creating some
charge separation, a dipole moment, thus carbon dioxide molecules can absorb IR
radiation. Collisions will immediately transfer this energy to heating the
surrounding gas. On the other hand, other CO2 molecules will be
vibrationally excited by collisions. Roughly 5% of CO2 molecules are
vibrationally excited at room temperature and it is this 5% that radiates. A
substantial part of the greenhouse effect due to carbon dioxide exists because
this vibration is easily excited by infrared radiation. CO2 has two
other vibrational modes. The symmetric stretch does not radiate, and the
asymmetric stretch is at too high a frequency to be effectively excited by
atmospheric temperature collisions, although it does contribute to absorption
of IR radiation. The vibrational modes of water are at too high energies to
effectively radiate, but do absorb higher frequency IR radiation. Water vapor
has a bent shape. It has a permanent dipole moment (the O atom end is electron
rich, and the H atoms electron poor) which means that IR light can be emitted
and absorbed during rotational transitions, and these transitions can also be
produced by collisional energy transfer. Clouds are also very important
infrared absorbers. Therefore, water has multiple effects on infrared radiation,
through its vapor phase and through its condensed phases. Other absorbers of
significance include methane, nitrous oxide and the chlorofluorocarbons.
Discussion of
the relative importance of different infrared absorbers is confused by the
overlap between the spectral lines due to different gases, widened by pressure
broadening. As a result, the absorption due to one gas cannot be thought of as
independent of the presence of other gases. One convenient approach is to
remove the chosen constituent, leaving all other absorbers, and the
temperatures, untouched, and monitoring the infrared radiation escaping to
space. The reduction in infrared absorption is then a measure of the importance
of that constituent. More precisely, define the greenhouse effect (GE) to be
the difference between the infrared radiation that the surface would radiate to
space if there were no atmosphere and the actual infrared radiation escaping to
space. Then compute the percentage reduction in GE when a constituent is
removed. The table below is computed by this method, using a particular
1-dimensional model of the atmosphere. More recent 3D computations lead to
similar results.
|
Gas removed
|
percent reduction in GE
|
|
H2O
CO2
O3
|
36%
12%
3%
|
By this
particular measure, water vapor can be thought of as providing 36% of the
greenhouse effect, and carbon dioxide 12%, but the effect of removal of both of
these constituents will be greater than 48%. An additional proviso is that
these numbers are computed holding the cloud distribution fixed. But removing
water vapor from the atmosphere while holding clouds fixed is not likely to be
physically relevant. In addition, the effects of a given gas are typically
nonlinear in the amount of that gas, since the absorption by the gas at one
level in the atmosphere can remove photons that would otherwise interact with
the gas at another altitude. The kinds of estimates presented in the table,
while often encountered in the controversies surrounding global warming, must
be treated with caution. Different estimates found in different sources
typically result from different definitions and do not reflect uncertainties in
the underlying radioactive transfer.
When Do You
Send Greenhouse Gases into the Air
Whenever
you...
Watch TVUse a
Hair Dryer
Use the Air
ConditionerRide in a Car
Turn on a
LightPlay a Video Game
Listen to a
StereoWash or Dry Clothes
Use a Dish
WasherMicrowave a Meal
... you are
helping to send greenhouse gas into the air.
To perform
many of these functions, you need to use electricity. Electricity comes from
power plants. Most power plants use coal and oil to make electricity. Burning
coal and oil produces greenhouse gases.
Other
things we do send greenhouse gases into the air
The trash that
we send to landfills produces a greenhouse gas called methane. Methane is also
produced by the animals we raise for dairy and meat products and when we take
coal out of the ground. Whenever we drive or ride in a car, we are adding
greenhouse gases to the atmosphere. And, when factories make the things that we
buy and use everyday, they too are sending greenhouse gases into the air.
And now
let’s talk about Climate and Weather
Weather is all
around us. Weather may be one of the first things you notice after you wake up.
Changes are, if it is cold and snowing, you'll wear a jacket when you go
outside. If it's hot and sunny, you may wear shorts. Sounds pretty simple,
right?
But what about
climate? How is it different from weather? And what is weather, exactly?
Weather
Weather
describes whatever is happening outdoors in a given place at a given time.
Weather is what happens from minute to minute. The weather can change a lot
within a very short time. For example, it may rain for an hour and then become
sunny and clear. Weather is what we hear about on the television news every
night. Weather includes daily changes in precipitation, barometric pressure,
temperature, and wind conditions in a given location.
Climate
Climate
describes the total of all weather occurring over a period of years in a given
place. This includes average weather conditions, regular weather sequences
(like winter, spring, summer, and fall), and special weather events (like
tornadoes and floods). Climate tells us what it's usually like in the place
where you live. San Diego is known as having a mild climate, New Orleans a
humid climate, Buffalo a snowy climate, and Seattle a rainy climate.
Is
the climate warming
An enhanced
greenhouse effect is expected to cause cooling in higher parts of the
atmosphere because the increased "blanketing" effect in the lower
atmosphere holds in more heat. Cooling of the lower stratosphere (about
30-35,000ft.) since 1979 is shown by both satellite Microwave Sounding Unit and
radiosonde data, but is larger in the radiosonde data.
There has been
a general, but not global, tendency toward reduced diurnal temperature range
(the difference between high and low daily temperatures) over about 50% of the
global land mass since the middle of the 20th century. Cloud cover has
increased in many of the areas with reduced diurnal temperature range.
Relatively
cool surface and tropospheric temperatures, and a relatively warmer lower
stratosphere, were observed in 1992 and 1993, following the 1991 eruption of
Mt. Pinatubo. The warming reappeared in 1994. A dramatic global warming, at least partly associated with the record El Nino, took place in 1998. This warming
episode is reflected from the surface to the top of the troposphere. Indirect
indicators of warming such as borehole temperatures, snow cover, and glacier
recession data, are in substantial agreement with the more direct indicators of
recent warmth.
Arctic sea ice has decreased
since 1973, when satellite measurements began but Antarctic sea ice may have
increased slightly.
Can we
change the climate
It may seem
hard to believe that people can actually change the Earth’s climate. But
scientists think that the things people do that send greenhouse gases into the
air are making our planet warmer.
Once, all
climate changes occurred naturally. However, during the Industrial Revolution,
we began altering our climate and environment through agricultural and
industrial practices. The Industrial Revolution was a time when people began
using machines to make life easier. It started more than 200 years ago and
changed the way humans live. Before the Industrial Revolution, human activity
released very few gases into the atmosphere, but now through population growth,
fossil fuel burning, and deforestation, we are affecting the mixture of gases
in the atmosphere.
Since the
Industrial Revolution, the need for energy to run machines has steadily
increased. Some energy, like the energy you need to do your homework, comes
from the food you eat. But other energy, like the energy that makes cars run
and much of the energy used to light and heat our homes, comes from fuels like
coal and oil – fossil fuels. Burning these fuels releases greenhouse gases.
Environmental
protection in Ukraine
In the 20th
century, the rapid growth of science and technology resulted in an increasing
negative effect on the biosphere of the Earth. Huge industrial enterprises
pollute the air we breathe? The water we drink and the land, which gives us
bread, vegetables, and fruit. Their discharge of dust and gas into the
atmosphere returns to the Earth in the form of acid rains. It also destroys the
ozone layer of the Earth and causes ‘’ greenhouse effect‘’. It effects forests,
rivers, crops and people’s health. This leads to the reduction of the life-span
of man. People die younger because of cancer, AIDS and other diseases which are
directly connected with the polluted environment they live in. Many species of
animals and birds face extinction due to the pollution of the biosphere.
The world’s
oceans are in danger too. They are filled with poisonous industrial and nuclear
waste, chemical fertilizers and pesticides. The Aral Sea in Russia is already
dead, the Mediterranean and the North Sea are slowly dying.
The worst
situation with air pollution is in big overpopulated cities. In Cairo and Mexico
City, for example, breathing is equivalent to smoking 2 packs of cigarettes a
day. The big industrial cities in Ukraine like Zaporizhiya, Donetsk, Kharkiv
and some others have the same situation.
Another threat
for the environment are nuclear power stations like Chernobyl. In April 1986
that nuclear power plant just north-west of Kyiv suffered the worst nuclear
accident in history: dozens died immediately, tens of thousands were evacuated,
while the long-term effects to human life are difficult to calculate. A large
part of Ukraine, Russia and Byelorussia was polluted by radioactive substances.
Great damage was done to their economy, nature and people’s health. The problem
of Chernobyl has not been solved yet because of the economic difficulties that Ukraine
is having now. The power plant was closed on December 15, 2000.
Nowadays
people of Ukraine, like most people in developed countries, realize that
without solving environmental problems, the life of the future generations will
be in real danger. Many people join the Great Party of Ukraine to unite their
efforts to save the planet where we live, to make our world healthier and more
beautiful.
Greenpeace
Greenpeace is
an international environmental organization founded in Vancouver, British
Columbia, Canada in 1971. It is best known for its campaigns against whaling.
In later years, the focus of the organization turned to other environmental
issues, including bottom trawling, global warming, ancient forest destruction,
nuclear power, and genetic engineering. Greenpeace has national and regional
offices in 42 countries worldwide, all of which are affiliated to the
Amsterdam-based Greenpeace International. The global organization receives its
income through the individual contributions of an estimated 2.8 million
financial supporters, as well as from grants from charitable foundations, but
does not accept funding from governments or corporations.
Mission
statement
Greenpeace's
official mission statement describes the organization and its aims thus:
Greenpeace is
an independent, campaigning organization which uses peaceful direct action and
creative communication to expose global environmental problems, and to force
solutions for a green and peaceful future. Greenpeace's goal is to ensure the
ability of the earth to nurture life in all its diversity.
Structure
Greenpeace is
a global environmental organization, consisting of Greenpeace International
(Stichting Greenpeace Council) in Amsterdam, and 27 national and regional
offices around the world, providing a presence in 41 countries. These national
and regional offices are largely autonomous in carrying out jointly agreed
global campaign strategies within the local context they operate in, and in
seeking the necessary financial support from donors to fund this work. National
and regional offices support a network of volunteer-run local groups. Local
groups participate in campaigns in their area, and mobilise for larger protests
and activities elsewhere. Millions of supporters who are not organized into
local groups support Greenpeace by making financial donations and participating
in campaigns as citizens and consumers.
National
and regional offices
Greenpeace is
present in the following countries and regions, as of March 2007: Argentina, Australia-Pacific region
(Australia, Fiji, Papua New-Guinea, Solomon Islands), Belgium, Brazil, Canada,
Chile, China, Czech Republic, France, Germany, Greenpeace Nordic (Denmark,
Finland, Norway, Sweden), Greece, Greenpeace Central and Eastern Europe
(Austria, Hungary, Slovak Republic, Poland, Romania, Bulgaria, Slovenia,
Serbia, Montenegro and Bosnia), India, Italy, Japan, Luxembourg, Greenpeace
Mediterranean (Israel, Cyprus, Lebanon, Malta, Tunisia, Turkey), Mexico, the
Netherlands, Greenpeace Aotearoa New Zealand (New Zealand), Russia, South-East
Asia (Philippines, Indonesia, Thailand), Spain, Switzerland, United Kingdom,
and the United States.
Friend of
Earth (FoE)
Friends of the
Earth is the U.S. voice of an influential, international network of grassroots
groups in 70 countries. Founded in San Francisco in 1969 by David Brower,
Friends of the Earth has for decades been at the forefront of high-profile
efforts to create a more healthy, just world. There members were the founders
of what is now the world's largest federation of democratically elected
environmental groups, Friends of the Earth International.
In March of
2005, Friends of the Earth finalized a merger with Bluewater Network. Bluewater
is a dynamic organization with creative campaigns to combat global warming, air
and water pollution and damage to public lands by thrill vehicles such as
snowmobiles and jetskis. The merger has added to our capacity and enabled us to
broaden the scope of our work in a number of areas.
Among there
present efforts are successes that draw headlines nationwide and international
and local efforts that make a difference in your backyard and those of people a
world away.
FoE conducted
lab tests that confirmed our suspicion that genetically engineered corn not
approved for human consumption was in products on supermarket shelves across
the nation.
They also
exposed the fact that Enron received $2.5 billion in taxpayer loans funneled
through international financial institutions.
In Indiana,
they are working with local groups to fight the destructive new-terrain I-69
project. This 140-mile, $1.8 billion highway would demolish thousands of acres
of farms and forests and bisect an Amish community.
Over the
years, there efforts and those of our supporters mean FoE have been able to:
stop over 150 bad dams and water projects worldwide; ban international whaling;
oust infamous James Watt; press for landmark regulations of strip mining and
oil tankers; reform the World Bank; and eliminate billions in taxpayer
subsidies to corporate polluters.
Literature
Internet data:
·
www.greenpeace.com
·
www.world-ecology.com
·
www.google.com.ua
Multimedia Editions
·
Britannica
Encyclopedia (Multimedia Edition)
·
British
Multimedia Encyclopedia
Books
·
Яблоков
А.В. Биология охраны природы.- Москва: Мир, 1983.-430 с.
·
Новиков
Г.А. Основы общей экологии и охраны природы. – Ленинград: Изд-во Ленингр.
ун-та, 1979.-352 с.
·
Никитин
Д.П., Новиков Ю.В. Окружающая середа и человек : Учебное пособие для студентов
вузов.- Москва : Высшая школа, 1986.- 415 с.
·
Меренюк
Г.В. Загрязнение окружающей среды и здоровье человека. – Кишинев : Штиница,
1984. – 144 с.
·
Дорогунцов
С.І., Коценко К.Ф., Аблова О.К. Екологія. – Київ : Либідь,1999.
·
Адаменко
О.М.,Косенко Я.В. Основи екології: Начальний посібник для вузів. – К.,2005.
·
Білявський
Г.О., Фурдуй Р.С., Костіков І.Ю. Основи екології: Підручник. – Київ : Либідь,
2005. – 408 с.
·
Тучина Н.В.,
Меркулова Т.К., Кузьмина В.С. Speak English with pleasure. – Харьков 2003,
с. 233
Размещено
на