Preparation of Cu-based oxygen carriers for Chemical-looping combustion
THESIS FOR THE DEGREE OF
BACHELOR OF CHEMICAL ENGINEERING
Preparation of Cu-based
oxygen carriers for Chemical-looping combustion
By Rosen Angelov: Prof. Dr.-Ing. Stefan Heinrich
Supervisors: Dipl.-Ing. Marvin Kramp.-Ing. Andreas Thon
Hamburg 2012
Contents
Introduction
. Theoretical part
.1 Capturing of CO2
1.2 Chemical Looping Combustion (CLC)
.3 Oxygen carriers in CLC process
.3.1 State of art
.3.2 General oxygen carriers characteristics
1.3.3 Dry impregnation method
1.4 Fluidization
.5 Fluidized Beds
.5.1 Advantages and disadvantages of the Fluidized-Bed
Reactor
.6 Objectives
. Experimental part
.1 Materials
.1.1 Gamma alumina
.1.2 Copper(II) nitrate trihydrate
.2 Preparing of solution
.3 Impregnation calculations
.4 Description of Fluidized bed reactor
.5 Description of process of impregnation
.6 Calcination
. Results
.1 SEM analyze of materials
3.1.1 SEM pictures at 250x zoom
3.1.2 SEM pictures at 1500x zoom
.1.3 Discussion of the SEM pictures
3.2 Fluidization of the materials. Calculating the
minimum fluidization velocity
.2.1 Puralox (ɣ-Al2O3)
.2.2 Impregnated/dried Al2O3/Cu(NO3)2
3.2.3 Al2O3/CuO used in CLC
facility
3.2.4 Discussion on fluidized tests
Introduction
days are generally accepted that the greenhouse gas in
developed industrial countries must be reduced as much as possible. Carbon
dioxide is one of the most important greenhouse gases contributing to global
warming. The CO2 capture and storage (CCS) is a process involving
the separation of CO2 and the storage over the long term. There are
different CCS technologies available or under development, but most of them are
consuming a lot of energy and cost a lot of money.
The original idea of Chemical-looping combustion with
using “solid oxidizing agents”, or as they are called now oxygen carriers, was
born back in 1954 by Warren Lewis and Edwin Gilliland. Their original idea was
producing pure CO2, which was free of inert gasses like nitrogen
[1]. CLC as a term was used for first time in 1987 by Ishida et al. [2]. The
next few years there are several publications in literature about CLC (Ishida
& Jin, 1994; Anheden, N<#"660341.files/image001.gif">
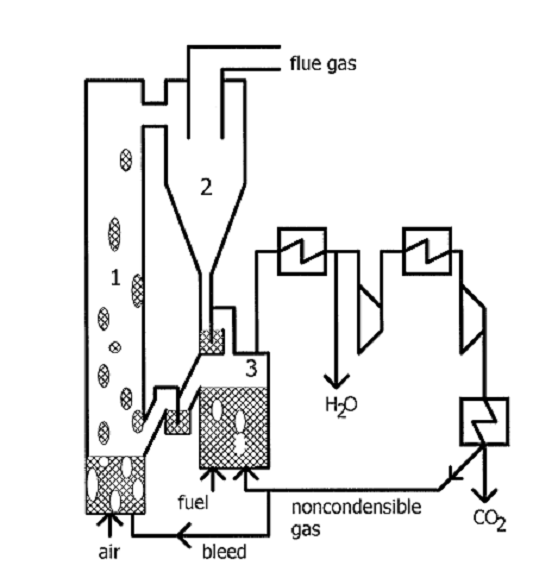
Figure 1. CCS Technology [5]
In the Post combustion fuel (gas/coal/biomass) and air
are mixed in power plant where inert N2 and unreacted O2 are
separated and almost pure CO2 is captured. Pre combustion the solid
fuel (coal or biomass) is gasificated with air flow and steam and then it went
through process of reforming where the separation of CO2 happened.
Reforming is followed by process of hydrogenation and then the fuel enters the
reactor. Exit gasses are N2 and O2.the oxyfuel process
air is separated in advance of inert nitrogen and oxygen. Oxygen and fuel are
mixed in reactor where the exit gas is CO2.are several possibilities
for such sequestration has been proposed [2]:
Ø storage in used oil and gas fields;
Ø storage in deep coal beds;
Ø storage in aquifers;
Ø deep sea storage;
Ø deep sea bottom storage. is other
useful option which includes some energy efficiency improvements, the switch of
less carbon-intensive fuels, renewable energy sources like sunlight and wind
power, nuclear power and others that must be considered [6].global growth and
distribution of CO2 emissions is given in figure 2 bellow.
 2. Global growth and distribution of
CO2 emissions 2000-2005 according to World Resources Institute [7]
2. Global growth and distribution of
CO2 emissions 2000-2005 according to World Resources Institute [7]
From this figure can be concluded that the most of released
emission are coming from electricity and heating, followed by transport and
industry. There is a trend of decreasing the amount of released CO2.
1.2 Chemical-looping Combustion
looping combustion is a novel technology for carbon
containing fuels preventing the CO2 emissions released at atmosphere
by inherent separation of the greenhouse gas carbon oxide. In the
chemical-looping combustion (CLC) process, fuel gas (natural gas, syngas,) is
burned in two interconnected reactors. In the first one, an oxygen carrier
(metal oxide) that is used as oxygen source is reduced by the feeding gas to a
lower oxidation state, where CO2 and steam are reaction products. In
the second reactor, the reduced solid is regenerated with air to the fresh
oxide, and the process can be repeated for over 100 successive cycles. The
carbon dioxide can be easily isolated from the outlet gas coming
from the fuel reactor by steam condensation [8].CLC system is made of two
interconnected reactors - air and fuel reactor, as shown in figure 3:

Fig. 3. Chemical-looping combustion. MeO/Me denote
recirculated oxygen carrier solid material.
the fuel reactor, the fuel gas is oxidized to CO2
and H2O by a metal oxide through the chemical reaction:
(2n + m)MeO + CnH2m → (2n + m)Me + mH2O
+ nCO2 (1)
exit gas stream from the fuel reactor contains CO2
and H2O, and almost pure CO2 is captured water is condense.
The reduced metal oxide, Me, is transferred into the air reactor where the
metal is oxidized according to equation (2):
+ ½O2 → MeO (2)
The flue gas leaving the air reactor contains N2
and unreacted O2. The exit gas from the fuel reactor contains CO2
and H2O, which are kept apart from the rest of the flue gas. After
water condensation, almost pure CO2 can be obtained with no energy
lost for component separation. Depending upon the metal oxide used, reaction
(1) is often endothermic, while reaction (2) exothermic. The total amount of
heat evolved from reactions (1) and (2) is the same as for normal combustion,
where the oxygen is in direct contact with the fuel [2].reactors in Fig. 2
could be designed in different ways, but two interconnected fluidized beds have
an advantage over other alternative designs, because the process requires a
good contact between gas and solids [1]. The system proposed is a circulating
system composed of two connected fluidized beds, a high-velocity riser and a
low-velocity bubbling fluidized bed (figure 4).the Chemical-looping combustion
the cornerstone are metal oxides which release oxygen - oxygen carriers. They
circulate between fuel and air reactor and fuel is never in direct contact with
air [9]. Oxygen carriers are first placed in air reactor. In air reactor the
fuel gas is oxidized by the metal oxides. The exhaust gases here are inert
nitrogen mostly and some unreacted oxygen. The driving force here is gas flow,
which makes particles in fluidized state. With increasing the gas flow rate,
velocity is increasing too.. Oxidized particles from air reactor through a
cyclone device are transferred in the fuel reactor. Here in the fuel reactor,
the fuel gas is reduced to CO2 and H2O. After water
condensation almost pure CO2 can be derived with minimum energy
lost. By-product here is ashes, which are result from the combustion. In
addition the fuel reactor can be considered as a bubbling fluidized bed and
particles from the fuel reactor are transferred to the air reactor back by the
force of gravity.
 4. Layout of chemical-looping
combustion process, with two interconnected fluidized beds [3].
4. Layout of chemical-looping
combustion process, with two interconnected fluidized beds [3].
- Air reactor
- Cyclone
- Fuel reactor
.3 Oxygen carriers in CLC process
1.3.1 State of art
This work was based on previous investigations. The dry
impregnation method technic was based on the investigation of Luis F. Diego et.
al [12]. He used CuO/Al2O3 oxygen carriers with content
of CuO between 10 and 26 wt %, prepared by wet and dry impregnation methods.
These oxygen carriers were analyzed in a fluidized bed facility during 100
reduction-oxidation cycles. He used CH4/N2 as fuel to
determine. It was found that CuO/Al2O3 oxygen carriers
with a CuO content lower than 10 wt % never agglomerated and those with a CuO
content greater than 20 wt % always agglomerate. It was concluded that this
phenomenon was indipendent of the preparation method and the calcination
temperature used in their muffle oven. On the other hand, the behaviour CuO/Al2O3
oxygen carriers with intermidiate CuO content (15-17 wt %) depended on
the calcination temperature used [12].oxygen carrier transfers oxygen from air
to the fuel, avoiding the direct contact between them. The metal oxide, used as
an oxygen carrier in chemical-looping combustion, must have sufficient
mechanical strength in multiple successive cycle reactions, good rates of
reduction and oxidation, must not agglomerate and must have pore structure. It
is also an advantage if the metal oxide is cheap and environmentally safe.
There are certain metals and their corresponding oxides which are proposed in
literature such as: Fe, Ni, Co, Cu, Mn, and Cd. These metals and their oxides
are combined with an inert that acts as a porous support enhancing the
reactivity properties and stability of the active phase. Moreover, this
inactive binder increases the mechanical strength and the attrition resistance
of the oxygen carrier. In the previous works, oxygen carriers based on Ni or Cu
show the highest reactivity with different support materials [10].
In the literature of chemical-looping combustion, the
conversion rate X is often used to determine the degree of conversion of the
oxygen carriers. The value is defined as the fraction of the difference between
the mass of the oxygen carrier (m) and the mass of the oxygen carrier in its
reduced state (mred), and the difference between the mass of the
oxygen carrier in its oxidized state (mox) and in its most reduced
state:

 (3)
(3)
degree of mass-based conversion ω is sometimes also used as a measure of the
oxygen carrier conversion, as it is convenient for comparisons of different
materials of different oxygen carrying capacity.
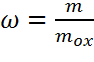
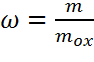 (4)
(4)
X can be converted to ω with the following equation:
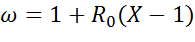
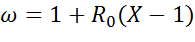 (5)
(5)
R0 is the oxygen transfer capacity, which
from the other side is the fraction of available oxygen in the oxygen carrier:

 (6)
(6)
There are several very important characteristics that
oxygen carriers must possess:
v Good mechanical strength.of the most
important qualities that the oxygen carriers must have is good mechanical
strength. This gives them sufficient durability in cycle reactions and lowers
the attrition rate of the particles during the CLC process.
v Good gas conversion.the efficiency of
the process sufficient gas conversion must be achieved in both fuel and air
reactors. This characteristic depends on several circumstances - type of used
oxygen carriers, support material, reactor type.
v Sufficient oxygen capacitymaximum
oxygen capacity is determined mainly by the type of oxygen carriers which were
used. For example in previous work where this property was investigated, it was
conclude that the oxygen capacity of the Cu- and Mn-based oxygen carriers have
the higher rates [11].
v High rates of reactionproper
selection of primary metal oxides, supports, particle synthesis techniques and
reaction conditions can enhance the reaction rate. Higher rates of reaction
even allow a small reactor can be used to achieve the same results like bigger
[12].
v Low cost priceraw material cost and
the cost of synthesizing the particles are of important economic consideration.
This must be taken on mind in case of large rates of production.
1.3.3 Dry impregnation methoddry impregnation method is often
used for preparing oxygen carriers for Chemical-looping combustion process. In
this work Cu-based oxygen carriers were prepared by this method and results
that appeared was concidered as satisfied. The first step is selection of the
support material. In that work material used as support was Puralox (ɣ-Al2O3).
The support is used in its powder form. Like it was mancioned in previous
chapter, support material improves the particles strenght and reactivity
abilities. This process were performed in fluidized bed reactor with
Wurster-draft tube. After being doped on the support, the oxygen carriers must
be calcined. Particles are first calcined at lower temperature for at least two
hours. The calcination results in reduction of the metal nitrates to metal
oxides. Finally, high-temperature calcination is carried out to obtain the
desired physical stability.. method is suitable for the synthesis of all types
of oxygen carriers. However the method predominantly is used for a copper-based
looping medium to reduce the effect of copper agglomeration. It is believed
that the reduced agglomeration effect results from limited metal loading in the
pores of the support [12].
1.4
Fluidization
Fluidization is process in which solid particles are
transformed into a fluidlike state through suspension in a gas or liquid [13].
The fluidization principle was first used on an industrial scale in
1922 for the gasification of fine-grained coal [14]. Since then, fluidized
beds have been applied in many industrially important processes. Fluidized beds
can be used for a large scale of processes such as cooling-heating, drying,
sublimation-desublimation, adsorption-desorption, coating, and granulation, to
many heterogeneous catalytic gas-phase reactions as well as noncatalytic
reactions [15].fluidization a packed bed of solid particles is brought to
a “fluidized” state by an upward stream of gas or liquid as soon as the
gas velocity rate of the fluid exceeds a certain limiting value Umf
(where mf denotes minimum fluidization). In the fluidized bed the
pressure drop ∆pfb of the fluid on passing through the fluidized
bed is equal to the weight of the solids minus the buoyancy, divided by the
cross-sectional area At of the fluidized-bed vessel (7):

 (7)
(7)
Equation (7), the porosity ε of the fluidized bed is the void
volume of the fluidized bed (volume in interstices between grains, not
including any pore volume in the interior of the particles) divided by the
total bed volume; s is the solids apparent density; and H is the height of the fluidized
bed. In many respects, the fluidized bed behaves like a liquid.
 5.
Pressure drop in flow through packed and fluidized bed
5.
Pressure drop in flow through packed and fluidized bed
1.5 Fluidized
Beds
oxygen impregnation alumina solution
The driving force of the fluidized bed reactors is the upward
flow of gas or liquids. There are different types of fluidized beds reactors,
depending of state that particles inside are. If the flow rate is very low and
the particles are still, then this bed is called Fixed (figure 6 a). When the
flow rate rise the bed come in the condition where the particles start rising a
bit and this bed is called Minimum fluidization bed (figure 6 b). If the flow
rate continues to rise, the process of forming bubbles has begun and this
bubbles move through the particles unitill they reach the surface of the bulk.
This is called Bubbling fluidized bed (figure 6 c). If the flow rate is to
high, the particles are carried out of the bed and there is no bulk formation.
In this case the fluidized bed is called Lean-phased (figure 6 e ).

 6. Forms of fluidized beds [15].
6. Forms of fluidized beds [15].
1.5.1 Advantages and disadvantages of the Fluidized-Bed
Reactor
There are several advantages of fluidized bed reactors that
were concidered during the experiments:
v The fluidized bed is easy to work
with
v The bad can be applied for particles
with different size
v Can be used for both small and large quantities
v There are no hot spots during the
process where the temperature is higher
v Some of the fluidized beds got
filters, that can be autocleanedare some disadvantages of fluidized beds:
v It can not be work on high
temperatures
v If the particles inside are with low
size, they can adhere to the fluidized bed shapes
v Some beds require specific software
1.6 Objectives
The main goal of this work was to synthesize large quantities of
Cu-based oxygen carriers with gamma alumina as support (ɣ-AL2O3),
which are used in Chemical-looping combustion (CLC) technology. The produced oxygen carriers should have
good qualities. The work in this thesis was mainly experimental. For the most
of the experiments was used fluidized bed reactor with Wurster-draft tube.
According to calculations before experiments, the produced Cu-based oxygen carriers
should have 13% content of CuO.
The other part of that work was to analyze the produced
material using Scanning Electron Microscope (SEM) and fluidization test
performed in fluidized bed reactor.
2. Experimental
2.1 Materials
For preparing of Cu-based oxygen carriers in this work were
used as support commercial ɣ-alumina (Puralox NWa-155,
Sasol, Germany GmbH) particles of 0.1-0.5 mm with a density of 0.76 g/cm3 and a
porosity of 55.4 % were used as support and dry impergnation method should be
applied. The properties of the materials used are given below.
2.1.1 Puralox
(ɣ- Al2O3)Al2O3 is
white fine white powder (see figure 7) used as support for Cu-based oxygen
carriers. It enhances the mechanical strength of the oxygen carriers and also
lowers the attrition rate. Gamma alumina has large specific surface (164 m2/g)
which made this support suitable for the purpose of this work. Other advantage
of this material is its relatively large porosity (54.5%). The higher porosity
allows more copper nitrate to be impregnated in the alumina particles. It is
not enviormentaly danger.
 7. Gamma alumina powder
7. Gamma alumina powder
Some of the material characteristics are given in Table 1 and
Table 2.
Table 1. Product information [16]
|
Synonyms
|
Gamma
alumina, Puralox NWa-155
|
|
Chemical
formula
|
ɣ- Al2O3
|
|
Molar mass
|
101.96 g/mol
|
2. Chemical and physical data [16]
|
Test
|
Units
|
Value
|
|
|
Specific
surface
|
m2/g
|
164.0
|
|
|
Al2O3
- content
|
%
|
98.4
|
|
|
SiO2
- content
|
ppm
|
51.0
|
|
|
Fe2O3
- content
|
ppm
|
54.0
|
|
|
Na2O
- content
|
ppm
|
4.0
|
|
|
Bulk density
|
g/cm3
|
0.76
|
|
|
Porosity
|
%
|
54.5
|
|
|
Particle
distribution <100 Mikron
|
%
|
1.0
|
%
|
99.9
|
|
|
Solubility
|
g/l
|
2670
|
distribution of the Puralox NWa-155 is shown in figure 8.
 8. Particles distribution of Puralox
NWa-155
8. Particles distribution of Puralox
NWa-155
2.1.2
Copper(II) nitrate trihydratecopper(II) nitrate trihydrate is small blue
crystals which are highly hygroscopic (figure 9). Left in contact with air they
agglomerate fast and become hard. The copper nitrate trihydrate has good
solubility in water. The measured solubility was 600 g/l and the solubility
given from the distributor is over four times higher (2670 g/l). The copper
nitrate has pH value 3-4, which makes it with acid characteristics.
 9. Copper(II) nitrate trihyrdate
crystals
9. Copper(II) nitrate trihyrdate
crystals
Some of the characteristics of copper nitrate trihydrate are
given in Tables 3 and 4.
|
Synonyms
|
Copper dinitrate
trihydrate
|
|
Chemical formula
|
Cu(NO3)2*3H2O
|
|
Molar mass
|
241.6 g/mol
|
Table 3. Product information [17]
|
Properties
|
Units
|
Value
|
|
Melting point
|
°C
|
114
|
|
Density
|
g/cm3(20°C)
|
2.05
|
|
Bulk density
|
kg/m3
|
1050
|
|
pH value
|
lgH+
|
3-4
|
|
Solubility
|
g/l (20⁰C)
|
2670
|
2.2
Preparing of solution
first part of experimental part was to prepare the oxygen
carriers. The first step was preparing solution of Cu(NO3)2*3H2O
for impregnation. Before making the solution, we need to investigate the
solubility of Cu(NO3)2*3H2O in water. To
investigate the solubility sample of 100g of the material was taken and placed
in the flask of glass, which weight is measured in advance. The flask was
placed on magnetic stirrer and small amounts of water were added during the
mixing process while the copper nitrate crystals are fully dissolved. After
it’s fully dissolved the flask is put on the balance. From the difference in
weights we can calculate the amount of water we used. The results from this
experiment are given in the table below:
5. Calculation of solubility
|
flask+fish, g
|
material, g
|
Material+water+flask,g
|
water only,g (18⁰C)
|
|
182,4
|
100
|
343
|
60.6
|
results are showing that 100g of Cu(NO3)2*3H2O
can be dissolved in 60.6g of water. Since solubility is known, the next step is
to prepare a solution in larger quantities. For this purpose is used larger
container and stirrer like ones shown in figure 10:
 10. Experimental setup for copper
nitrate solution
10. Experimental setup for copper
nitrate solution
When the exact amount of solution need and proportions
material/water are calculated, first the water was placed in container, and
then while stirrer is working on small portions was added the copper nitrate.
After about hour of mixing the copper nitrate was dissolved and the solution
was ready. The used copper nitrate trihydrate was not with sufficient quality
and after dissolving of the solution in the bottom of the container there was
white sludge left. Then optimization of method was tried like using the
gravitational sedimentation and pumping the upper layer with limpid solution in
a 10 litter tube. The final solution can be seen in figure bellow:

Figure 11. Solution of Cu(NO3)2*3H2O
2.3
Impregnation calculations
For calculating how much solution was needed for exact amount
of alumina, the following equations were used:
) Finding the mass of CuO needed. We know in advance
that the copper oxide in the material should be 13% of total weight.
mCuO = mAl2O3*0.13, g
) Calculating the mole fraction of CuO:
nCuO = mCuO/MCuO, mol
where MCuO is molar mass of copper oxide and it
is:
MCuO = MCu + MO, g/mol
)
Calculating
the mass of the solid crystals Cu(NO3)2*3H2O:
m[Cu(NO3)2*3H2O] = M[Cu(NO3)2*3H2O]*n[Cu(NO3)2*3H2O],
g
From the mole equation the followed conclusion was made:
n[Cu(NO3)2*3H2O]:n[CuO] =
1:1
) Calculating the amount of water needed to dissolve
the solid material:
mH2O = (m[Cu(NO3)2*3H2O]*60.6)/100,
g
These values come from the conclusion, that 100g of the
copper nitrate trihydrate are dissolved in 60g of water.
) Calculating the final amount of solution
msolution = m[Cu(NO3)2*3H2O]
+ mH2O, g
2.4 Description of the fluidized bed reactor
 11. Technical scheme of Fluidized bed
11. Technical scheme of Fluidized bed
2.5
Description of impregnation process
The next step after the solution was ready was dry
impregnation (discussed in 3.3.3) performed in fluidized bed reactor shown in
figure 12. The alumina powder is placed in the Wurster-Coating cylinder shown
in figure 12, which has a perforated bottom with applied filters. These filters
retain large and fine particles, and also allow air flow through them. The
alumina powder during the process must not outflow through them. The average
amount of alumina used for one cycle of process was 4.1 kg.
 12. Wurster-Coating cylinder
12. Wurster-Coating cylinder
After measuring the exact amount of alumina was putted in the
cylindrical part, the fluidized bed was hermetically sealed and the process was
ready to begin. First was needed to set the air flow rate, which determines how
high the bulk would rise. With the higher flow rate set, the particle will
reach higher.fluidized bed got heater that was set to 40°C over the process of
spraying. Peristaltic pump (figure 13) was connected to the bed via hose and
automatically controlled by the system software according needed rate.
 13. Peristaltic pump
13. Peristaltic pump
rate of the pump was set to 45g/min. After solution was
pumped, it was sprayed over the cylindrical part from the nozzle in the middle.
The process of spraying can be seen in figure 14:
 14. Spraying process in
Wurster-Coater fluidized bed reactor
14. Spraying process in
Wurster-Coater fluidized bed reactor
During the process of spraying the moisture of the material
was changing with the amount of solution was sprayed because of the adsorption
of the material. next step after the whole amount of solution was sprayed was
to dry the particles. For the drying process the temperature of the heater was
set to 80°C. The material was dry when the moisture remained.
2.6 Calcination
Calcination involves heating the particle to temperatures
close to or higher than the sintering temperature for an extended period of
time. The calcination procedure was carried out under an air atmosphere. Therefore,
metal salts in the particle such as metal nitrates will be converted to metal
oxides according to equation (0). For this purpose the oxygen carriers were put
for at least two hours in muffle oven at 550 °C. The resulting, poisonous
nitrogen dioxide must be removed. After over two hours at this temperature, the
sample of oxygen carriers looks like these in figure 15.

 15. Oxygen carriers calcined at 550°C Figure 16. Oxygen carriers
calcined at 850°C
15. Oxygen carriers calcined at 550°C Figure 16. Oxygen carriers
calcined at 850°C
calcination at 550°C a typical blackening for copper was
spotted. Some of the particles went from light blue color to green blue and
this was indication that hydroxide still content in the oxygen carriers.next
step was calcination at 950°C for at least two hours again. The color of the
oxygen carriers can be seen in figure 16. After second calcination they were
brighter with a brown shade
3. Results
.1 SEM images of materials
For analyzing the surface of the materials, JEOL 840a
scanning electron microscope (figure 16) was used to make the SEM pictures.
 16. JEOL 840a SEM
16. JEOL 840a SEM
Before working on SEM, the probes were processed with
Au(gold) film for 40 seconds at 40kV. That step was made for better SEM
pictures. Gold particles cover the probes and as is known the gold is the best
metal conductor. Covering of the particles with this film improves
significantly the quality of the taken pictures.better analysis of the probes,
four materials will be observed together at different zoom and compared.
3.1.1 SEM pictures at 250x zoom




A - Puralox NWa-155 (ɣ- Al2O3)-
Impregnated/dried Al2O3/Cu(NO3)2-
Al2O3/CuO used in CLC facility- Al2O3/CuO
calcined at 850⁰C
3.1.2
SEM pictures at 1500x zoom




A - Puralox NWa-155 (ɣ- Al2O3)-
Impregnated/dried Al2O3/Cu(NO3)2-
Al2O3/CuO used in CLC facility- Al2O3/CuO
calcined at 850⁰C
3.1.3
Discussion of the SEM picturesshowed that alumina particles, which were not
involved in any process, have round shape (A). Their surface is smooth and
there are no sharp edges. After the impregnation of the material there are
crystals of CuO on the surface of alumina particles (B). These crystals are
distributed evenly over the surface. That can be sign for successive
impregnation of the fresh material, even the desired result is impregnation in
the inner part of alumina. the pictures of Al2O3/CuO used
in CLC facility can be observed that the particles are deformed from the
attrition, which fluidization process caused (C). There are sharp edges and the
surface is almost flat. Particles calcined at 850⁰C shown indications for
CuO crystals on their surface, but these crystals are significantly less than
fresh impregnated one.
3.2
Fluidization of the materials. Calculating the minimum fluidization velocity umf
fluidization tests were performed in small fluidized bed
shown in figure 17 below:
 17. Fluidized bed for testing umf
17. Fluidized bed for testing umf
were made during different pressure by two different
controllers.
3.2.1
Puralox (ɣ-Al2O3)

calculated minimum fluidization velocity umf for ɣ-Al2O3 is:
|
Material
|
Umf, cm3/s
|
ρbulk, kg/m3
|
|
Puralox Nwa-155
|
1,60
|
760
|
3.2.2 Impregnated/dried Al2O3/Cu(NO3)2

calculated minimum fluidization velocity umf for Impregnated/dried Al2O3/Cu(NO3)2 is:
|
Material
|
Umf, cm3/s
|
ρbulk, kg/m3
|
|
Al2O3/Cu(NO3)2 impregnated
|
2,32
|
1064,9
|
.2.3 Al2O3/CuO used in CLC facility

The calculated minimum fluidization velocity umf
for Al2O3/CuO used in CLC facility is:
|
Material
|
Umf, cm3/s
|
ρbulk, kg/m3
|
|
Al2O3/CuO used in CLC facility
|
2,42
|
939,5
|
3.2.4
Discussion of fluidized testsminimum fluidization velocity raised gradually.
The results were compared with previous works [18], which shown normal fluidization
behavior of the particles.
Conclusions
Based on previous investigations a large scale method for
Cu-based oxygen carriers preparation was established by dry impregnation
method. During the preparation of spraying solution it was observed the
formation of a white precipitate, which is a sign that the material used is not
pure.
It was conclude that the fluidized bed reactor with
Wurster-draft tube show good results as example a large amount of material (4,2
- 4,4 kg) can be prepared on one cycle. Also using of the Wursted-Coater has
advantages because of the position of spraying nozzle, which is in the bottom
side and it is in the bed of particles. This allows good contact, sufficient
efficiency, between particles and sprayed solution compared with previous works
where the nozzle was above the particles and the nozzle often blocked. It is
also was concluded that the particles did not agglomerate during the process.
An amount of 180 kilograms Cu-based oxygen carriers with
Puralox as support with 13% content of CuO was prepared. After calcination at
550°C a typical blackening for copper was spotted. Some of the particles went
from light blue color to green blue and this was indication that hydroxide
still content in the oxygen carriers. Sample of the material after calcination
lose 18,9 % of its weight because of evaporation of the water molecules. After
the calcination at 850°C material went light brown from almost black.of the
Prualox, impregnated Al2O3/Cu(NO3)2,
Al2O3/CuO used in CLC facility and Al2O3/CuO
calcined at 850°C were analyzed using Scanning Electron Microscope (SEM)
pictures. In these pictures can be seen that the surface of the impregnated Al2O3/Cu(NO3)2
content equally distributed crystals of CuO, which is sign for successive
impregnation. After calcination in CLC facility the surface of particles was
flat most is inside.same samples beside Al2O3/CuO
calcined at 850°C were put to fluidization test performed in fluidized bed
reactor. The minimum fluidization velocity were calculated and compared with
results from previous works, which shown normal fluidization behavior of the
particles.
Acknowledgements
of all I want to thank Prof. Dr.-Ing.
Stefan Heinrich for the given opportunity to do my thesis in Hamburg University of
Technology (TUHH). Also I would not succeed without the support and help of my supervisors
Dipl.-Ing. Marvin Kramp and Dipl.-Ing. Andreas Thon. I want to thank the entire
department “Solid Processes Engineering and Particle Technology” which helped
me in my work: Dr.
Ernst-Ulrich Hartge, Dipl.-Ing. Miika Franck, Heiko
Rohde and all
the rest. Last but not least I want to thank the Bulgarian practicant Vassil Traychev for helping me
with the experimental work.
References
[1] Lewis, W.K. and E.R. Gilliland. (1954 USA): Production
of pure carbon dioxide, US patent no: 2665971
[2] Ishida, M., D. Zheng, and T. Akehata, Evaluation
of a chemical-looping combustion power-generation system by graphic exergy
analysis, Energy (Oxford, United Kingdom), 1987, 12(2): 147-54.
[3] Lyngfelt,A., Leckner, B., Mattisson, T. (2
December 2000): A fluidized-bed combustion process with inherent CO2
separation; application of chemical-looping combustion, Department of Energy
Conversion, Chalmers University of Technology, 412 96 Goteborg, Sweden
[4] Adanez, J., Abad, A., Garcia-Labiano, F., Gayan,
P., de Diego, F. (2011): Progress in Chemical-Looping Combustion and Reforming
technologies, Progress in Energy and Combustion Science 38 (2012) 215e282
[5] http://www.kbr.com/Images/Content/Full/CO2-Capture-Processes.jpg
[6] IPCC Working Group III, (Montreal, Canada, 22-24
September 2005): Carbon Dioxide Capture and Storage
[7] World Resources Institute (2005)
http://www.wri.org/files/wri/images/emissions-growth.preview.png
[8] Adanez, J., Garcfa-Labiano, F., de Diego, L.F.,
Gayan, P., Abad, A. and Celaya, J. (2005): Carbon Dioxide Capture for Storage
in Deep Geologic Formations, Volume 1, D.C. Thomas and S.M. Benson (Eds.)
[9] Jerndal, E., Mattisson, T., Thijs, I., Snijkers,
F., Lyngfelt, A. (2010): Investigation of NiO/NiAl2O4 oxygen carriers for
chemical-looping combustionproduced by spray-drying
[10] Adanez, J., Garcfa-Labiano, F., de Diego, L.F.,
Gayan, P., Abad, A., Celaya, J. (2005): Greenhouse Gas Control Technologies,
Volume I, Instituto
de Carboquímica (ICB-CSIC), Dept. of Energy
& Environment, Miguel Luesma Castán, 4, Zaragoza, 50018, Spain
[11] De Diego, L., Gayan, P.,
Garcia-Labiano, F., Celaya, J., Abad, A., Adanez, J. (2005): Impregnated CuO/Al2O3
Oxygen Carriers for Chemical-Looping Combustion: Avoiding Fluidized Bed
Agglomeration, Energy & Fuels 2005, 19, 1850-1856
[12] Gupta, P., Li, F., Velazquez-Vargas, L., Sridhar,
D., Iyer, M., Ramkumar, S., Fan, L.-S. (2005): CHEMICAL LOOPING SYSTEMS FOR
FOSSIL ENERGY CONVERSIONS
[13] Levenspiel, O., Kunii, D. (1991): Fluidization
Engineering second edition, copyright by Butterworth-Heinemann
[15] Werther, J., Hamburg University of Technology,
Hamburg, Germany (2007): Fluidized-Bed Reactors
[16] Sasol, Germany
[17] Merck KgaA, Frankfurter Str. 250, 64293
Darmstadt, Germany
[18] Leion, H., Chalmers University of Technology Göteborg, Sweden 2008“
Capture of CO from
Solid Fuels using Chemical-Looping Combustion and Chemical-Looping with Oxygen
Uncoupling”